INDIA has started what authorities claim is the world's fastest and largest inoculation drive, with 191,000 healthcare workers receiving a dose of a COVID-19 vaccine on Saturday.
health
Jan. 17, 2021
STAFF REPORTER
7 min read
India starts massive COVID-19 inoculation drive
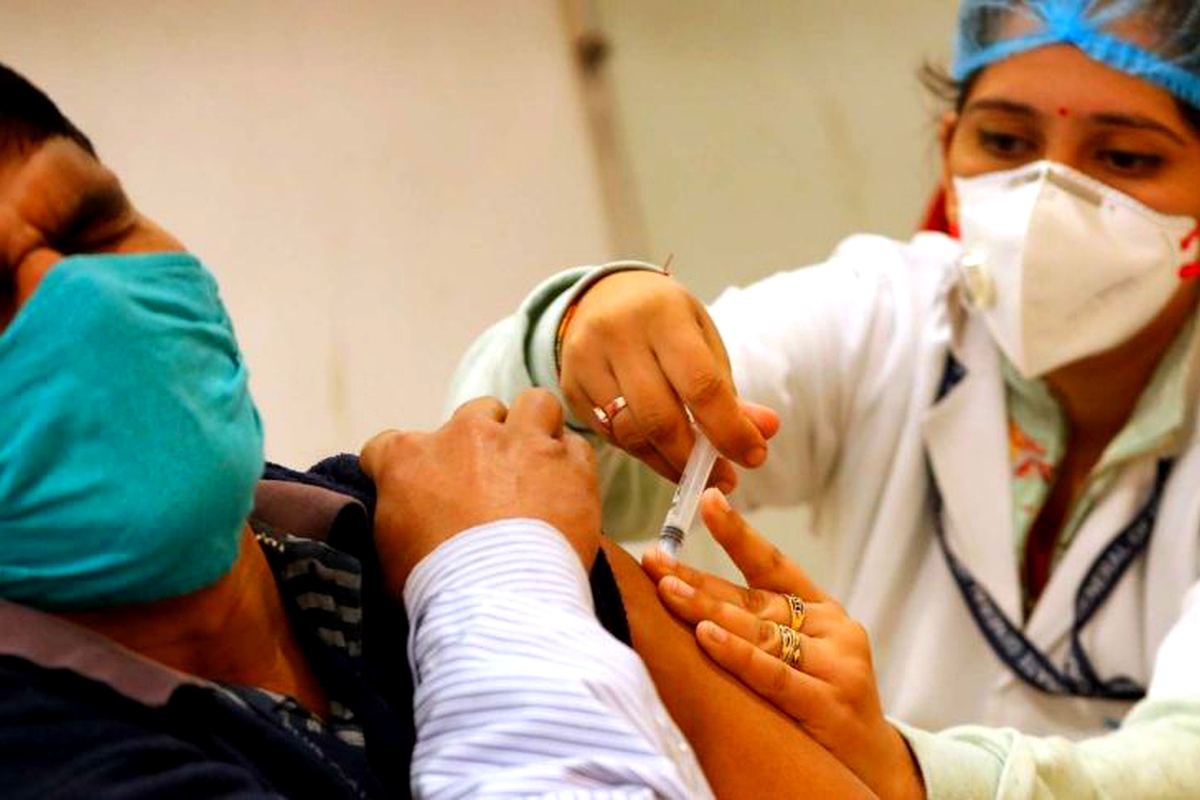
An Indian health worker administers a Covid-19 vaccine to a patient on Saturday
The Government's first phase of an ambitious vaccination program will target 30 million people, starting with healthcare and frontline workers before it is expanded to people aged above 50 and those suffering from comorbidities.
Authorities have set up an initial 3,000 vaccination points across India, each to provide doses to 100 people a day.
By August, India aims to have inoculated 300 million people.
Indian Prime Minister Narendra Modi launched the vaccine rollout in a televised address, stating the world was "looking at India with hope".
"Such a vaccination drive at such a massive scale has never been conducted in history," he said.
"This disease kept people away from their families. People could not meet their elderly admitted at hospitals. We could not bid adieu to those with proper rituals who died due to corona."
Despite the newfound optimism, Mr Modi warned people to not let their guard down, coining the phrase "vaccine as well as discipline".
"Start of vaccination does not mean people should stop following the COVID protocols of wearing masks and maintaining social distancing," he said.
Health expert Suneela Garg from the Maulana Azad Medical College — who is helping the capital Delhi with its vaccine program — said the huge population of India would present some challenges.
"It's a mammoth task," Dr Garg told the ABC.
"We have the population of many countries. But we are all prepared."
'Getting vaccinated is the answer to ending this'
India is home to 1.36 billion people and has recorded more than 10.5 million COVID-19 cases, the second-highest in the world, and 151,000 deaths.
The Country was experiencing almost 100 000 daily cases during its peak in September last year but has enjoyed a mostly steady downward trend in recent months — with daily infections having dropped below 16,000.
Farah Husain, who manages intensive care wards in Delhi's largest coronavirus hospital, Lok Nayak Hospital, said the vaccine would provide “an armour” for her and other frontline workers.
"There's a huge sense of relief and a hope," Dr Husain said.
Dr Husain said she signed up to be one of the first to be inoculated for two main reasons.
One was that it would prevent her from catching the virus, and the other was that it would also stop her from potentially spreading it to her elderly parents and grandparents who live nearby.
"It's one step towards their safety," she said.
"And that's how the entire world should see it. Getting vaccinated is the answer to ending this COVID disease."
Dr Husain's 67-year-old father, Aniq Husain, is a former cardiac and hypertension patient and hopes to get the vaccine in two or three months.
"I'm highly thrilled," he said.
"Because the past one year, it was like a jail and now I feel like we may be able to come out of the prison."
Her 92-year-old grandfather, Faiq Husain, enjoys long walks outside to maintain his fitness but has had to take ample precautions due to his age and heart problems.
"It's been very, very challenging to live a normal life," he said.
Patients won't get to choose which vaccine they get
The inoculation program has been a moment of national pride as the two vaccines given emergency approval are made in India.
The first is the Oxford-AstraZenneca vaccine, known locally as Covishield, which is being manufactured by the world's largest vaccine manufacturer, the Serum Institute, based in Pune.
The second is Covaxin, which was developed by local pharmaceutical company Bharat Biotech.
Tens of millions of doses were transported across the country in the days before the rollout began, with each vaccine relatively cheap and easy to store.
Those taking part in the initial phases of the vaccine rollout will not be able to choose their preferred vaccine, and each injection site will stock only one brand of vaccine to avoid confusion.
However, the two vaccines have attracted controversy ahead of their rollout.
Authorities hoped to inoculate 300,000 frontline workers on the first day, but local media is reporting some hesitation among workers led to the shortfall.
Like Australia, there is debate in India about the efficacy and appropriate dosage of the Oxford-AstraZeneca vaccine.
Meanwhile, Covaxin was approved before the completion of phase 3 trials, prompting concern about data transparency.
Health experts have questioned why the authorities did not wait a few weeks for the trials to be finished, but Dr Garg said she was confident Covaxin was safe and effective.
"We have observed, in the phase 1, this is a very safe vaccine," she said.
"We found 92 per cent and 98 per cent levels of immunoassay were there in phase 2."
Some challenges lie ahead
The vaccine rollout is anticipated to be opened up to the private market around the middle of the year.
Health experts say efforts to inoculate 300 million people won't be enough to achieve "herd immunity" but it will protect the vulnerable and prevent deaths.
Epidemiologist Giridhar Babu from the Public Health Foundation of India said finding vulnerable people would be challenging given India's huge population and geographical diversity.
Enjoy our daily newsletter from today
Access exclusive newsletters, along with previews of new media releases.

He said efforts to identify those at risk should be made immediately while the number of cases was relatively low.
"The government health workers and the government frontline workers are much easier to [vaccinate] because there is complete data on them," Dr Babu said.
"The tougher part is to vaccinate persons with comorbidities and the elderly.
"The elderly is a bit easier because you can get it from the census data. For those with comorbidities, the data is not complete."
Will COVID-19 vaccinate drive undermine other efforts?
India spends a paltry 1.29 per cent of GDP on public healthcare, compared to more than 9 per cent in the UK and Australia.
There are concerns the COVID-19 inoculation drive will undermine other important health and immunisation programs.
For example, health experts suspect the number of tuberculosis cases last year halved because detection equipment was diverted to help the fight against COVID-19.
India has also postponed its 2021 polio vaccination drive, citing "unforeseen circumstances".
"In programs like the [tuberculosis] elimination program, some of the gains that have been done over decades might be reversed," Dr Babu warned.
Dr Garg said Delhi had already drafted plans to ensure that does not happen.
"COVID will not be our only attention," Dr Garg said.
"We are only going to utilise only half of the manpower for COVID-19 vaccination program, the rest of them will carry out the activates of immunisation … related to different programs." BBC News
Tailored for you






